Our research aims to offer novel solutions to prevent and treat such diseases. For example, we are developing novel materials to reduce biofouling of surfaces, with an emphasis on design of protein and bacteria resistant coatings. A common theme in this research area is the use of mussel-inspired strategies for linking antifouling polymers onto surfaces. Specifically, we often employ the adhesive qualities of DOPA or catechol containing molecules for grafting antifouling polymers onto surfaces (Figure). These coatings can be made by adsorbing catechol modified polymers onto a surface, or by adsorbing an initiator and growing a bound polymer by surface-initiated polymerization.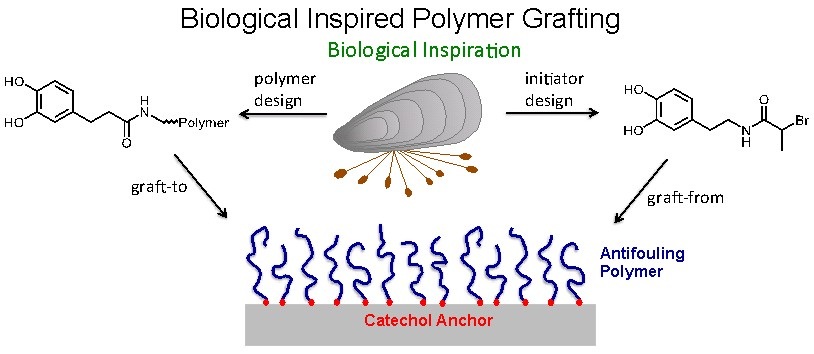
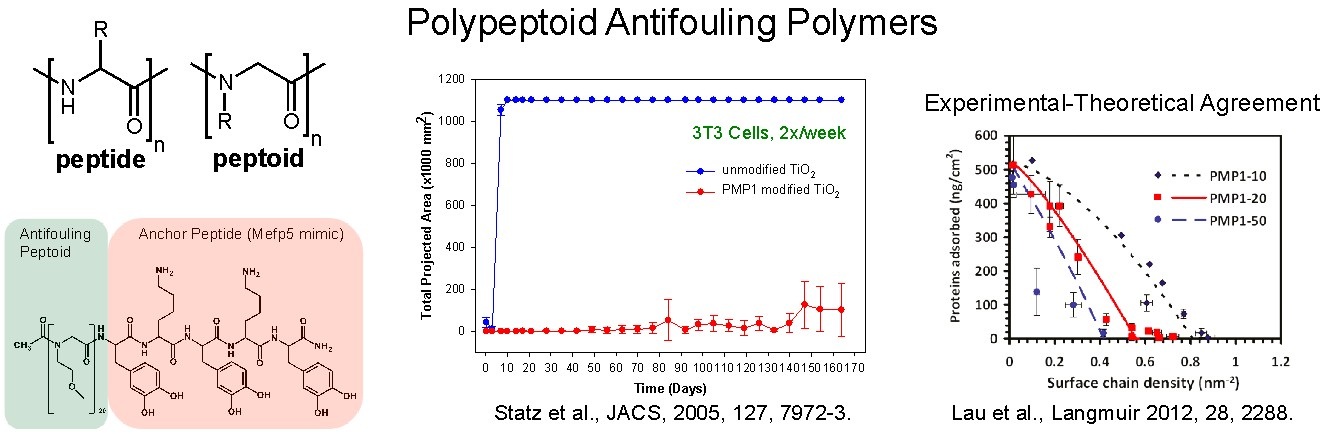
We were the first group to employ catechols as polymer anchors, adsorbing the antifouling polymer PEG onto oxide surfaces through DOPA containing peptides (JACS, 2003 and Langmuir 2005). More recently, we have pioneered the development of novel antifouling surface-grafted poly(N-substituted glycine) (peptoids) as antifouling polymers. These polymers are synthesized by solid phase synthesis for the convenient incorporation of a diverse range of sidechains and for precise monomer-by-monomer control over residue sequence and chain length. Surface grafted peptoid brushes provide excellent resistance against non-specific protein adsorption and mammalian and bacterial cell attachment (Figure). Furthermore, the convenient and precise synthetic control afforded by peptoids has enabled the systematic study of important phenomena regarding the rational design of antifouling surfaces in terms of both the chemistry and surface architecture. Specific experiments have also been designed in collaboration with the theory group of Professor Igal Szleifer for the integrated analysis of experimental results with molecular theory and molecular dynamics simulations, to provide powerful insight into the molecular mechanisms governing antifouling behavior.